Introduction
Ac-225 is an α-emitting radionuclide with a half-life of 9.9 days. Its high energy, multi-day half-life and short travel distance in tissues makes it an attractive radioisotope for targeted radiotherapy (TRT) for cancers. Currently, there are about 15 ongoing clinical trials worldwide in prostate cancer, leukemia and micrometastatic carcinomas that underscore the potential of Ac-225 based radiotherapies.
There are no Ac-225 therapies currently approved but approvals are expected beginning 2027-2028. Current demand for Ac-225 in the marketplace is for clinical trials and pre-clinical R&D work. About 1000 patients are being recruited for Ac-225 trials globally in clinical trials with an estimated demand of about 27 GBq (~ 700 mCi) of Ac-225 (ref.)
Ac-225 in Targeted Radiotherapy
Manufacture of Ac-225 from thorium-229 (Th-229) generators is a traditional method for obtaining Ac-225. Ac-225, typically available as Ac-225 nitrate or Ac-225 chloride, is converted to a TRT drug in an automated synthesizer, with general structure as shown in Figure 1.
Sourcing Th-229
One of the major suppliers of Ac-225 via the Th-229 Generator route is Oak Ridge National Laboratory (ORNL), TN, USA. FDA approved ORNL’s Type II Drug Master File (DMF) for actinium-225 nitrate from Th-229 in April 2021. Figure 2 shows the parent radioisotope Th-229 is isloated from, and decay of Th-229 to Ac-225.
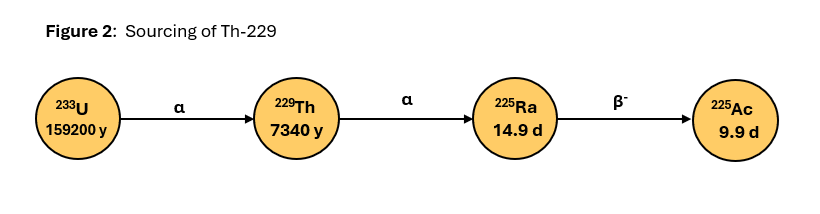
As shown in Figure 2, Th-229 is sourced from uranium-233 (U-233). U-233 is stored at ORNL and is from the nuclear stockpile from 1950-1960. This material is highly regulated and its storage and disposal is managed by the US Dept. of Energy (DOE). Due to the extremely long half-life U-233 to decay to Th-229 (159,200 years), quantities of Th-229 are extremely limited.
Isotek is the contractor company tasked by DOE for isolation and purification of Th-229 from U-233 stored at ORNL under “Thorium Express” project started in 2019. To-date, Isotek has isolated about 15 grams of Th-229 (and expects to extract additional 40 g through the end of project) (Ref. ). The “small” amount of Th-229 isolated shows the complexity of handling the highly radioactive waste and complex and time consuming processing.
Although 15 g of Th-229 extracted does not sound much, it is substantially higher, ~ 1500%, compared to worldwide inventory of Th-229 (~ 1 g) outside of ORNL!
Ac‑225 isolation begins by treating U‑233 stock with nitric acid, converting radiometal compounds into a solution of uranyl, actinium, thorium, and other impurity metal nitrates. This mixture is passed through a series of ion exchange columns to isolate Th‑229, then purified and further processed to obtain Ac‑225.
Activity Calculations
The activity of Ac-225 at equilibrium that could be obtained from 15 g of Th-229 can be estimated; the calculations for the same are shown in Figure 3:
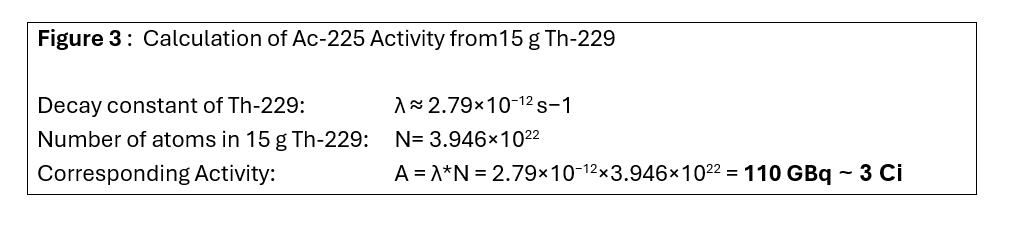
The calculations show that the activity is ~ 110 GBq, about 3 Ci, a relatively small amount; this amount is ~ three times the estimated Ac-225 requirement estimated for worldwide clinical trials.
As previously noted, ORNL produces Ac‑225 using a process that is documented in its FDA-accepted DMF. ORNL also has an agreement with TerraPower Isotopes (TPI), Bellevue, WA , and supplies extracted Th-229 to TPI for the manufacture of Ac-225. According to its website, TPI began supplying Ac‑225 for clinical trials on a weekly basis from October 2024 (Ref). TPI also offers research-grade (non-GMP) Ac‑225 for use in R&D applications.
Isolation and purification of Ac-225
A general scheme for the isolation of Ac-225 from U-233 is shown in Figure 4 below.
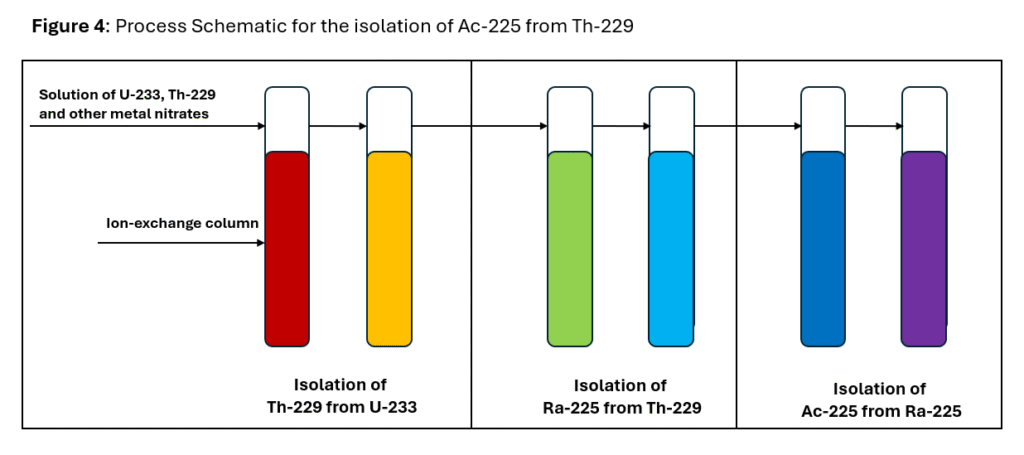
The process involves isolation of Th‑229 from U-233, Ra‑225 from Th-229 and Ac-225 from Ra-225. Ac-225 is further purified and is typically supplied as a nitrate or chloride as noted earlier.
Unlike Ga‑68 or Tc‑99m generators, Ac-225 generators are not amenable for on-site elution at hospitals or small-scale facilities due to the complexity of the process. However, given Ac-225’s nearly 10-day half-life, it can be efficiently distributed from a centralized supplier, with manageable activity loss during ground or air transport of up to 2 days, as an example.
There are other suppliers that manufacture Ac-225 via the Th-229 route. They are:
PanTera, Belgium, supplying Ac-225 since June 2025 (Th-229 is supplied by TPI) and Cardinal Health, in collaboration with TPI, provide cGMP grade Ac-225 weekly “at scale” per CH website.
Conclusion
Producing Ac‑225 using Th‑229 generators represents the conventional method of Ac-225 manufacture and currently supports ongoing clinical research and development. This generator-based approach is helping to bridge the supply gap for Ac‑225 while alternative production methods such as cyclotron-based generation using Ra‑226 are in development. Given its reliability and scalability, the Th‑229 route is expected to continue playing a vital role in meeting the near-term demand for Ac-225 in the growing field of targeted radiotherapeutics.
